天麻(Gastrodia elata Bl.)为兰科多年生草本植物,常以其干燥块茎入药,具息风止痉、平抑肝阳之功效[1],2023年11月被正式列入《按照传统既是食品又是中药材的物质目录》[2]。天麻的功能性成分包括酚类、甾醇、有机酸及多糖等,其中以天麻素(gastrodin,GAS)、对羟基苯甲醇(p-hydroxybenzyl alcohol,HBA)和巴利森苷(Parishin)类物质最为关键,天麻素是酚苷类化合物,对羟基苯甲醇是酚类化合物,巴利森苷类是复杂酚苷类化合物,它们都属于天麻的主要活性成分且在代谢和结构上存在递进关系[3]。天麻素及其代谢产物对羟基苯甲醇(苷元)共同发挥神经保护、镇静催眠及抗氧化作用,巴利森苷类物质是一系列由天麻素、对羟基苯甲醇及有机酸通过酯键连接而成的天然化合物群,通过缓慢释放活性单元实现抗炎与免疫调节的协同增效[4]。然而,天然状态下这些成分普遍存在生物利用度低、血脑屏障穿透性差、代谢不稳定等问题[5],因此,富集核心活性成分是实现功能性食品标准化的前提。
天麻活性成分当前主要提取方法包括水/醇回流提取结合大孔树脂纯化[6],辅以超声或微波辅助提升效率[7];超临界萃取与膜分离技术用于高纯组分制备[8]。为进一步突破体内转化瓶颈,提高生物利用度,微生物发酵成为关键策略,微生物发酵通过代谢调控与酶解转化生成有益次级代谢产物,不仅能显著改善天麻的特殊气味,增强活性成分含量及健康功效,还有助于提升其安全性[9-10]。酵母菌与乳酸菌分泌的β-葡萄糖苷酶与酯酶可高效水解天麻素与巴利森苷类物质,定向释放游离对羟基苯甲醇,显著提升其生物可利用性与中枢靶向性[11];相较于化学法(副反应多、结构破坏)与粗酶法(杂酶干扰),微生物体系兼具反应温和、高效、安全等优势,符合“药食同源”产品的绿色开发导向[12-13]。研究天麻微生物发酵转化技术路径,对推动天麻从传统药材向高稳定、高吸收、高适口的功能性食品转型具有重要意义。
为系统解析天麻核心活性成分的结构、功能与机制,深入探究微生物关键酶系对天麻主要酚类活性物质定向转化的调控机制,筛选定向转化的产酶菌株并构建多菌协同体系,本文总结了天麻主要酚类活性成分(天麻素、对羟基苯甲醇和巴利森苷类)的药理活性及其代谢关联性,揭示了天麻素向对羟基苯甲醇转化的核心药效路径及巴利森苷类的“储库”缓释特性;构建了由β-葡萄糖苷酶与酯酶协同驱动的多级酶联催化网络,阐明其从巴利森苷前体到活性对羟基苯甲醇的定向转化机制;梳理黑曲霉(Aspergillus niger)、里氏木霉(Trichodermareesei)、枯草芽孢杆菌(Bacillus subtilis)、马克斯克鲁维酵母(Kluyveromyces marxianus)、植物乳植杆菌(Lactiplantibacillus plantarum)等高效产酶菌株,为高附加值天麻功能性制品开发提供理论支撑。
1 天麻中主要酚类活性成分及其功效
1.1 天麻素
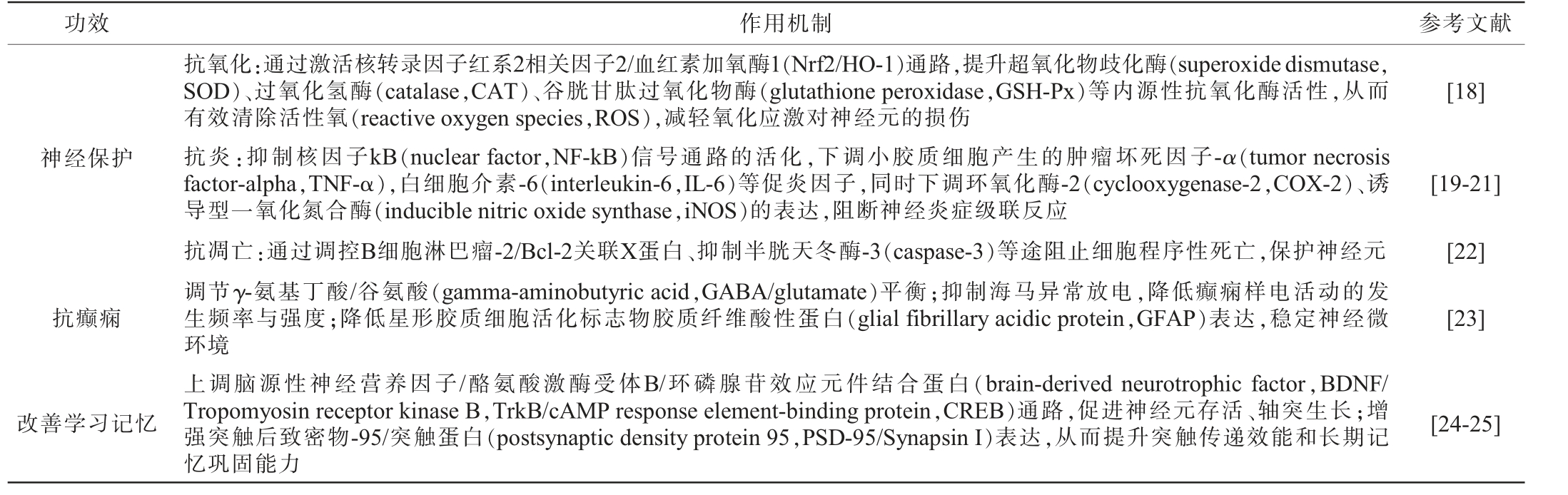
天麻素属于酚苷类化合物,由一个对羟基苯甲醇(苷元)与一个葡萄糖通过β-糖苷键连接而成,化学名为4-羟甲基苯基-β-D-吡喃葡萄糖苷,分子式为C13H18O7。天麻素是天麻中活性成分含量最高的有效单体成分,是天麻的主要质控指标之一[14],在天麻中天麻素的含量通常在0.1%~1.0%之间,因品种、产地、加工方式等因素而异。云贵地区天麻中天麻素含量高于其他地区,可达0.6%[15],红天麻中天麻素、巴利森苷类含量通常高于乌天麻[16]。此外,减压干燥和冷冻干燥有利于保持天麻素含量[17]。天麻素水溶性好,脂溶性差,热稳定性中等,在酶作用下易水解为对羟基苯甲醇(HBA),巴利森苷类在酸/酶作用下可水解生成天麻素[3]。其主要功效包括神经保护、抗癫痫、改善学习记忆,其作用机制见表1。
表1 天麻素主要功效及其作用机制
Table 1 Main pharmacological effects and mechanisms of Gastrodia elata
1.2 对羟基苯甲醇
对羟基苯甲醇属于小分子酚类化合物,化学名为4-羟基苯甲醇,是天麻素的苷元,无糖基修饰,极性较低,易溶于乙醇、乙醚,水中溶解度较低,较天麻素(GAS)更易透过生物膜[26]。它通常以游离形式存在于植物体内,对羟基苯甲醇在天麻不同部位中含量分布不均,其中块茎中含量最高,种子中次之,而在天麻杆、叶和蒴果壳中均未检出[27]。天麻中对羟基苯甲醇天然状态下含量较低,通常在0.01%~0.20%范围内,却是发挥药效活性的关键物质[15]。该物质具有显著的血脑屏障保护作用,其主要功效为神经保护、抗血小板聚集、扩张脑血管,其作用机制详见表2。
表2 对羟基苯甲醇主要功效及其作用机制
Table 2 Main pharmacological effects and mechanisms of p-hydroxybenzyl alcohol

1.3 巴利森苷
巴利森苷类化合物为天麻特有成分,是以天麻素及其衍生物在不同羧基位点与柠檬酸缩合形成的酚类成分,主要根据化学结构差异分为巴利森苷A(Parishin A)、巴利森苷B(Parishin B)、巴利森苷C(Parishin C)、巴利森苷E(Parishin E)等30多种成分[35],分子质量高、脂溶性差,口服吸收困难,需依赖酶解逐步释放活性物质HBA、GAS。其功能特性主要包括神经保护、免疫调节、抗疲劳,作用机制详见表3。
表3 巴利森苷类主要功能特性及作用机制
Table 3 Main pharmacological effects and mechanisms of parishins

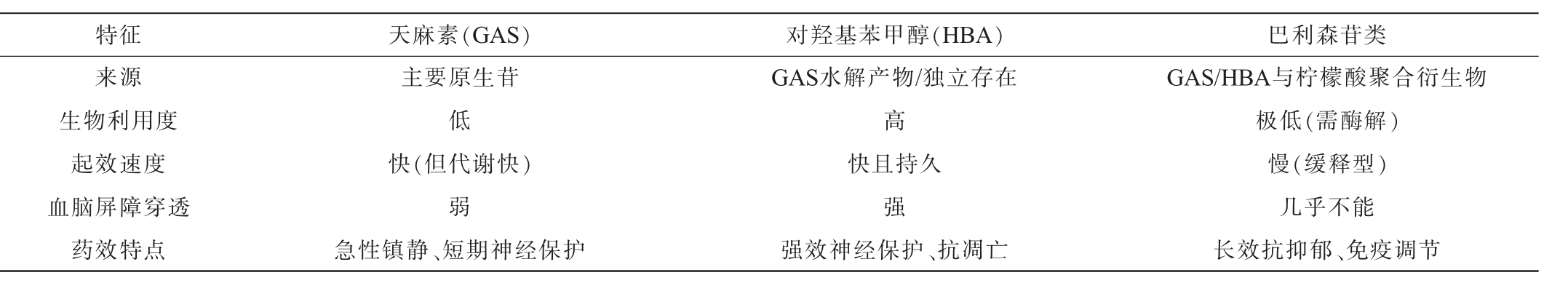
2 主要酚类活性成分药效动力学特征
天麻主要酚类活性成分药效动力学特征见表4。由表4可知,天麻素是天麻中的主要初生苷,口服后易被肠内水解,生物半衰期短,体内稳定性差、靶向递送效率低,严重制约其神经保护等中枢药效的发挥,亟需通过结构修饰或生物转化策略提升其药代动力学性能;HBA被认为是天麻素体内发挥药效的“真正活性形式”,其药理强度常高于化合物GAS[43];巴利森苷类虽含量高(占天麻干质量1%~3%),但因结构复杂、吸收差,其体内药效高度依赖生物转化[44]。因此,天麻提高生物利用度的核心在于巴利森苷及天麻素向HBA的高效转化。
表4 天麻主要酚类活性成分药效动力学特征
Table 4 Pharmacodynamic characteristics of major phenolic bioactive compounds in Gastrodia elata
3 酶对天麻主要酚类活性成分的转化
3.1 酶对天麻主要酚类活性成分的转化路径
酶对天麻主要酚类活性成分的转化路径见图1。
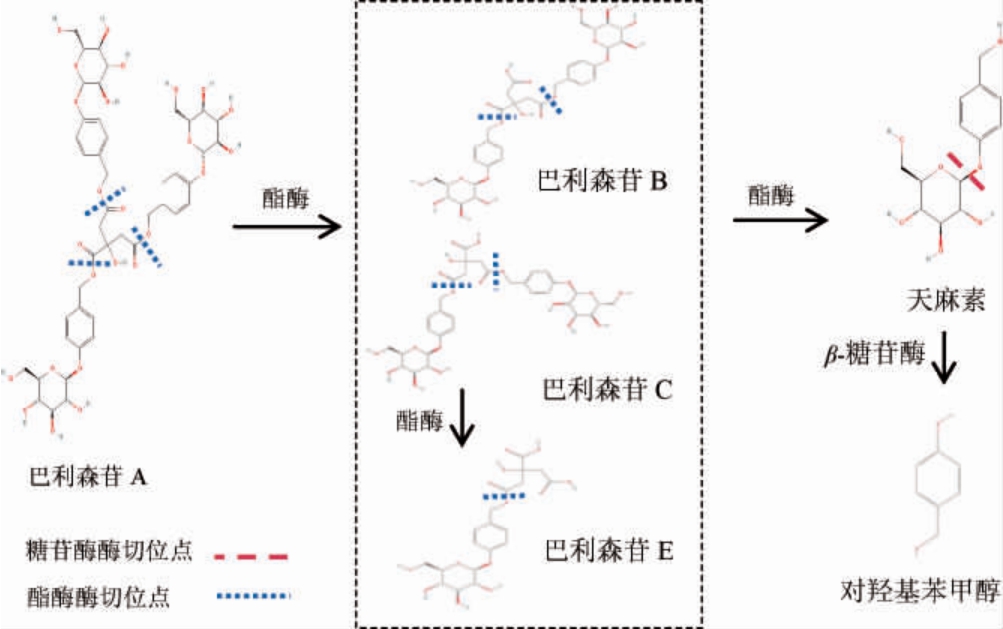
图1 酶对天麻主要酚类活性成分的转化路径
Fig.1 Transformation pathways of main phenolic bioactive compounds in Gastrodia elata by enzyme
由图1可知,巴利森苷A(Parishin A)在酯酶(esterase)作用下转化巴利森苷B(Parishin B)、巴利森苷C(Parishin C),进一步转化成巴利森苷E(Parishin E);Parishin B、Parishin C、Parishin E在酯酶作用下进一步转化为天麻素(GAS)[45-46],而GAS经β-葡萄糖苷酶水解“葡萄糖-酚羟基”之间的β-1,4-糖苷键,释放游离HBA[47-48]。形成巴利森苷(Parishins)→天麻素(GAS)→对羟基苯甲醇(HBA)的“串联水解”路径。
3.2 天麻主要酚类转化活性成分的酶解机制
天麻主要酚类转化活性成分水解酶的酶解机制及其酶解效果见表5。由表5可知,天麻主要酚类活性成分水解酶主要包括β-葡萄糖苷酶(Ⅰ型、Ⅱ型、Ⅲ型)、酯酶。β-葡萄糖苷酶(尤以Ⅰ型为代表)通过保留型双置换机制,特异性水解天麻素中的β-糖苷键,高效释放神经活性分子HBA;而酯酶(特别是高选择性酚酯水解酶)则通过丝氨酸三联体催化机制,精准切断巴利森苷中的酚-柠檬酸酯键,一步生成HBA,避免中间产物积累。两者分别承担“苷元释放”、“前体激活”功能,通过微生物共发酵实现在时空维度上的协同催化,显著提升HBA等核心活性成分的产率与生物可利用性。β-葡萄糖苷酶依据底物特异性可分为Ⅰ型、Ⅱ型、Ⅲ型,Ⅰ型芳香基特异性β-葡萄糖苷酶凭借其对天麻素底物的高亲和力与专一性,高效水解糖苷键生成对羟基苯甲醇,成为定向转化的核心酶;相比之下,Ⅱ型纤维二糖水解酶对天麻素无活性,Ⅲ型广谱β-葡萄糖苷酶则因催化效率低且副反应多而不适于精准转化。酯酶是串联反应的关键启动者,通用型酯酶可非特异性打开“糖/酚-柠檬酸”酯键,生成天麻素中间体;而高选择性的特异性酚酯水解酶则能精准识别对“羟基苯甲醇-柠檬酸”酯键,直接释放活性单元对羟基苯甲醇,具体催化机制及酶解效果详见表5。
表5 天麻主要酚类活性成分水解酶的酶解机制及其酶解效果
Table 5 Enzymatic hydrolysis mechanism and effect of hydrolase for main phenolic bioactive compounds in Gastrodia elata

4 天麻主要酚类活性成分转化的产酶菌株及其应用效果
4.1 产β-糖苷酶菌株的酶学特性及其应用效果
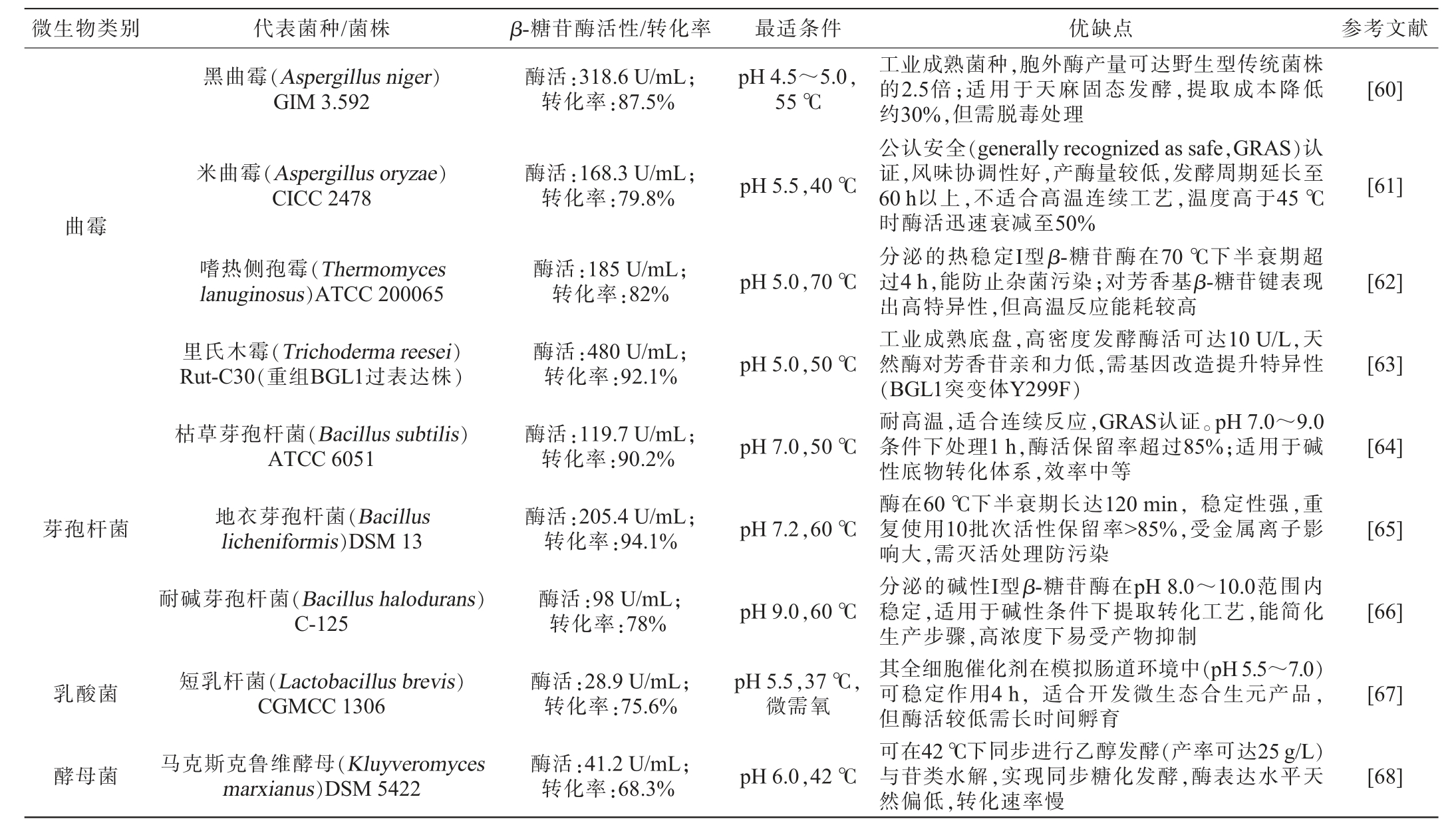
天麻素虽然是天麻的标志性成分,但大量的药理研究已证实,对羟基苯甲醇才是天麻在中枢神经系统发挥药效的“最终执行者”[26]。为实现天麻活性成分向对羟基苯甲醇的高效转化,筛选能够分泌高活性β-糖苷酶的微生物是关键。β-糖苷酶广泛存在于细菌、真菌和放线菌中[58]。研究显示,土壤、植物根际、发酵食品(如泡菜、豆豉)、动物肠道及药用植物共生微生物是高潜力菌源。部分极端环境(如温泉、酸性矿坑)来源菌株亦表现出耐热或耐酸特性,适合工业应用[59]。目前对已筛选出的高产β-葡萄糖苷酶代表菌株的产酶特性及其应用优缺点进行总结(表6)。
表6 产β-糖苷酶菌株的酶学特性与应用优缺点
Table 6 Enzymatic properties and application advantages and disadvantages of β-glucosidase-producing strains
续表

由表6可知,它们在催化效率和应用上各有优势:黑曲霉(Aspergillus niger)GIM 3.592所分泌的耐酸β-糖苷酶对巴利森苷表现出较高的底物亲和力[60];米曲霉(Aspergillus oryzae)CICC 2478作为公认安全(GRAS)的微生物,其酶制剂在药品生产中具有明确的合规性优势[61];嗜热侧孢霉(Thermomyces lanuginosus)ATCC 200065可分泌热稳定I型β-糖苷酶[62];里氏木霉(Trichoderma reesei)Rut-C30(重组BGL1过表达株)是最具规模化潜力的工程菌,适合GAS转化为HBA的大规模连续生产[63];枯草芽孢杆菌(Bacillus subtilis)ATCC 6051所产的碱性β-糖苷酶对多种酶抑制剂具有良好耐受性[64];地衣芽孢杆菌(Bacillus licheniformis)DSM 13经重组表达获得的热稳定性β-糖苷酶适用于固定化生物反应器工艺[65];耐碱芽孢杆菌(Bacillus halodurans)C-125可产碱性稳定性β-糖苷酶[66];短乳杆菌(Lactobacillus brevis)CGMCC 1306在厌氧条件下进行全细胞催化可有效抑制副产物的生成[67];马克斯克鲁维酵母(Kluyveromyces marxianus)DSM 5422所产的耐热酶系适用于萃取耦合的反应体系[68];巴斯德毕赤酵母(Pichia pastoris)GS115工程菌株则通过高密度发酵实现了酶蛋白的高效异源表达[69]。
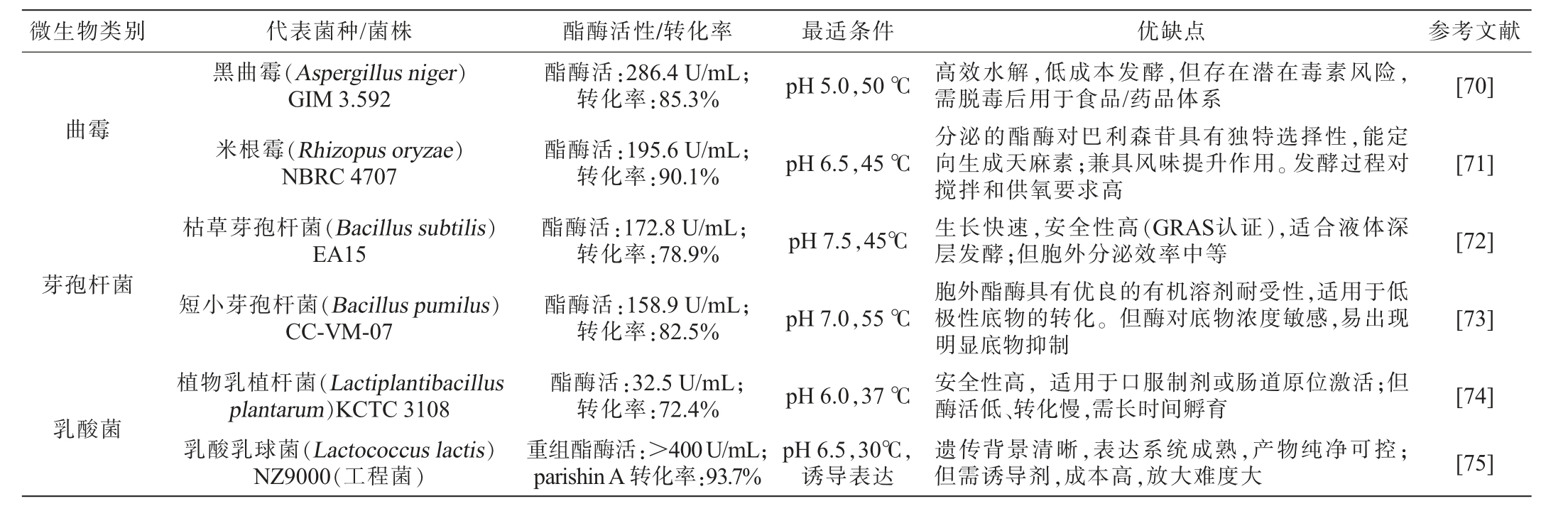
4.2 产酯酶菌株的酶学特性及其应用效果
在天麻研究中“酯酶”主要用于水解巴利森苷类成分。目前对已筛选出的高产酯酶代表菌株的产酶特性及应用优缺点进行总结,结果见表7。由表7可知,黑曲霉(Aspergillus niger)GIM 3.592表现出卓越的胞外酯酶合成能力,其对巴利森苷分子中酯键的水解效率显著,但存在潜在毒素风险,需脱毒后用于食品/药品体系[70]。米根霉(Rhizopus oryzae)NBRC 4707具有高选择性,能定向酶解生成天麻素,兼具风味提升作用[71];枯草芽孢杆菌(Bacillus subtilis)EA15所分泌的碱性酯酶在温和反应条件下即可实现对巴利森苷的高效生物转化[72]。短小芽孢杆菌(Bacilluspumilus)CC-VM-07具有优良的有机溶剂耐受性,适用于低极性底物转化[73]。植物乳植杆菌(Lactiplantibacillus plantarum)KCTC 3108作为公认安全(GRAS)的菌株,在医药与功能性食品领域具有明确的应用前景[74]。通过基因工程改造的乳酸乳球菌(Lactococcus lactis)NZ9000工程菌,其重组酯酶的催化效率得到实质性提升,展现出优异的工业化开发价值[75]。
表7 产酯酶菌株的酶学特性与应用优缺点
Table 7 Enzymatic properties and application advantages and disadvantages of esterase-producing strains
5 结论与展望
本文系统阐释了天麻核心活性成分(天麻素、对羟基苯甲醇、巴利森苷类)的功效及作用机制,并深入探究了基于β-葡萄糖苷酶与酯酶的转化路径机制及酶解机制,构建了由β-葡萄糖苷酶与酯酶协同驱动的多酶串联催化网络,实现了活性成分的高效释放与风味同步重构,梳理了多种高产β-糖苷酶与酯酶菌株的酶学特性与转化效率。然而,天麻微生物发酵的产业化进程仍受制于多菌种协同体系的稳定性不足、产物风味与转化效率难以兼顾以及酶解成本偏高等关键瓶颈。未来研究应聚焦于:①利用代谢工程精准调控双酶共表达工程菌的时序性表达,以替代不稳定的天然菌群;②借助多组学技术与人工智能,理性设计与创制催化效率更高、适应性更强的多酶复合体;③开发集成数字孪生与在线传感技术的智能化发酵系统,实现工艺参数的自适应优化与产品质量的精准控制;④推进绿色制造理念,通过原料全利用与能源循环,构建环境友好的可持续生产模式。
[1] SUBRAMANIAM S,WEN X Y,JING P.One-step microwave curingdehydration of Gastrodia elata Blume:Relationship between phytochemicals,water states and morphometric characteristics[J]. Ind Crops Prod,2020,153:112579.
[2] 市场监管总局,国家卫生健康委,国家中医药局.关于发布《按照传统既是食品又是中药材的物质目录(2023年版)》的公告[Z].2023年第42号,2023.
[3] CHEN C,SHI J.Advances in the study of phenolic acids and their pharmacological effects on the central nervous system in Gastrodia elata[J].Ind Crops Prod,2019,140:111585.
[4] 张志龙,郜玉钢,臧埔,等.天麻素、对羟基苯甲醇对中枢神经系统作用机制研究进展[J].中国中药杂志,2020,45(2):312-320.
[5] 龚其海,石京山,杨丹莉,等.天麻素在中枢神经系统的药理作用及其机制[J].中国新药与临床杂志,2011,30(3):176-179.
[6] 田梦凡,高青青,易雅丽,等.超声辅助酶提取、双水相萃取及大孔树脂纯化天麻素的工艺研究[J].中医药导报,2024,30(2):53-58.
[7] SELVAKUMAR P,SIVASHANMUGAM P.Ultrasound assisted oleaginous yeast lipid extraction and garbage lipase catalyzed transesterification for enhanced biodiesel production[J].Energ Convers Manage,2018,179:141-151.
[8] FAN W D,YING Y P,PEH S B,et al.Multivariate polycrystalline metalorganic framework membranes for CO2/CH4 separation[J]. J Am Chem Soc,2021,143(42):17716-17723.
[9] HU Y,ZHANG L,WEN R,et al.Role of lactic acid bacteria in flavor development in traditional Chinese fermented foods:A review[J].Food Res Int,2022,156:111373.
[10] ZHAO M,ZHANG T,WANG J,et al.Lactic acid fermentation modifies the volatile profile and reduces off-flavor compounds in traditional Chinese medicinal herb extracts[J]. LWT-Food Sci Technol,2023,178:114721.
[11] LIU Y,LIANG X,WANG H,et al.Biotransformation of gastrodin to p-hydroxybenzyl alcohol by Saccharomyces cerevisiae:Enzyme kinetics and pathway analysis[J].J Agr Food Chem,2021,69(12):3780-3788.
[12] 皋斌,徐丝瑜,鱼红闪.白头翁皂苷糖基水解Ⅰ型酶的酶学性质及特异性研究[J].中国生物工程杂志,2025,45(10):36-51.
[13] ABDELLA A,EL-BAZ A F,IBRAHIM I A,et al.Biotransformation of soy flour isoflavones by Aspergillus niger NRRL 3122 B-glucosidase enzyme[J].Nat Prod Res,2017,32(20):2382-2391.
[14] YANG H,WANG J,CHEN Y,et al.Comprehensive review of chemical constituents and pharmacological mechanisms of Gastrodia elata[J].Front Pharmacol,2023,14:1245678.
[15] 严国,蔡亚,杨易,等.新鲜天麻中天麻素与对羟基苯甲醇含量及影响因素分析[J].食品工业科技,2021,42(6):233-240.
[16] 周媛,周娜,王泽旭,等.安徽金寨红天麻、红乌天麻高效液相色谱法指纹图谱与化学成分比较[J].安徽医药,2023,27(4):683-690.
[17] ZHANG W,ZHENG S,WANG Y,et al.Effects of different drying and processing methods on the quality of Gastrodia elata[J].J Ethnopharmacol,2023,303:115962.
[18] ZHANG J Q,LI L Y,LIU Q,et al.Gastrodin programs an Arg-1+microglial phenotype in hippocampus to ameliorate depression-and anxietylike behaviors via the Nrf2 pathway in mice[J]. Phytomedicine,2023,108:31-45.
[19] CHEN L,WANG X,LIU M,et al.Anti-neuroinflammatory mechanisms of gastrodin in LPS-activated microglia: New insights[J]. Neuropharmacology,2022,200:109168.
[20] ZHAO Y,ZHANG Y,LIU H,et al.Gastrodin inhibits expression of iNOS and COX-2 in microglia[J].Biochem Biophys Res Commun,2014,450(4):1397-1402.
[21] WANG H,SUN Y,ZHANG L,et al.Gastrodin attenuates neuroinflammation through inhibition of microglial MAPK/NF-kB pathways[J].Eur J Pharmacol,2021,906:174221.
[22] LI Y,WU Q,CHEN J,et al.Gastrodin protects dopaminergic neurons via anti-excitotoxicity in Parkinson's models[J]. ACS Chem Neurosci,2020,11(10):1475-1485.
[23] WANG H,SUN Y,ZHANG L,et al.Gastrodin alleviates seizure severity and neuronal excitotoxicities in the rat lithium-pilocarpine model of temporal lobe epilepsy via enhancing GABAergic transmission[J]. Eur J Pharmacol,2021,906:174221.
[24] ZENG Y Q,GU J H,CHEN L,et al.Gastrodin as a multi-target protective compound reverses learning memory deficits and AD-like pathology in APP/PS1 transgenic mice[J].Front Pharmacol,2021,12:685946.
[25] FASINA O B,WANG J Y,MO J X,et al.Gastrodin from Gastrodia elata enhances cognitive function and neuroprotection of AD mice via the regulation of gut microbiota composition and inhibition of neuron inflammation[J].Front Pharmacol,2022,14:924123.
[26] XIAO T,YU X Z,TAO J,et al.Metabolomics-based study of the protective effect of 4-hydroxybenzyl alcohol on ischemic astrocytes[J]. Int J Mol Sci,2024,25(18):9907.
[27] 段昊,闫文杰.天麻辅助改善记忆保健功能的研究进展[J].食品科技,2023,48(3):72-79.
[28] SHA W,RONG H,RUIQIN Z,et al.Gastrodin and gastrodigenin improve energy metabolism disorders and mitochondrial dysfunction to antagonize vascular dementia[J].Molecules,2023,28(6):2598.
[29] ZHAO M H,ZHOU Y T,SHENG R L,et al.Gastrodin relieves Parkinson's disease-related motor deficits by facilitating the MEK-dependent VMAT2 to maintain dopamine homeostasis[J]. Phytomedicine,2024,132:155819.
[30] LIN Y E,LIN C H,HO E P,et al.Decision letter: Glial Nrf2 signaling mediates the neuroprotection exerted by Gastrodia elata Blume in Lrrk2-G2019S Parkinson's disease[J].Elife,2021,10:e73753.
[31] HAN M,LIU J,ZHANG H,et al.Novel antiplatelet and antithrombotic effects of natural compounds:Mechanisms and therapeutic potential[J].Phytomedicine,2023,120:154872.
[32] ZHANG R,WANG L,CHEN Y,et al.Anti-inflammatory and antiplatelet activities of phenolic compounds from medicinal plants[J]. J Ethnopharmacol,2022,287:115034.
[33] LIU Y,WANG X,CHEN J,et al.Vasodilatory effects of natural phenolic compounds via NO-cGMP pathway in vascular disease[J].Front Pharmacol,2023,14:1123456.
[34] CHEN H,WANG J,LIU Y,et al.Endothelium-dependent vasorelaxation mechanisms of plant-derived compounds[J].Vasc Pharmacol,2022,158:107123.
[35] 张菊,宋娜丽,马克坚.天麻中巴利森苷类成分药理作用、体内过程研究进展[J].中成药,2022,44(7):2223-2229.
[36] NING J,YAO C,ZHANG Y,et al.Antidepressant effects of Parishin C in chronic social defeat stress-induced depressive mice[J]. J Ethnopharmacol,2024,325:117891.
[37] ZHANG R,WANG L,CHEN Y,et al.Antidepressant mechanism of Parishin B in chronic stress model[J]. J Ethnopharmacol,2021,279:114445.
[38] WANG L,ZHAO W,LIU Y,et al.Immunomodulatory effects of plantderived compounds via TLR4 signaling pathway[J].Int Immunopharmacol,2023,112:109876.
[39]ZHANG W,LIU J,CHEN Y,et al.Immunomodulatory effects of bioactive compounds on splenocytes function[J].J Ethnopharmacol,2022,295:115678.
[40] WANG L,XU J,QIN L,et al.Parishins enhance mitochondrial function under hypoxia[J].J Ethnopharmacol,2020,261:113542.
[41] LUO Y,ZHANG X,WANG H,et al.Exercise performance enhancement through natural compounds: mechanisms and applications[J].J Funct Foods,2023,105:105562.
[42] ZHANG L,WANG Y,LIU J,et al.Gut microbiota-mediated metabolism of natural compounds: Pharmacokinetic implications and therapeutic potential[J].Pharmacol Res,2022,176:106141.
[43] HUANG J,LIU Y,WANG Y,et al.Intervention of gastrodin in type 2 diabetes and its mechanism[J].Front Pharmacol,2021,12:710722.
[44] ZHANG R,WANG L,CHEN Y,et al.Antidepressant mechanism of parishin B in chronic stress model[J]. J Ethnopharmacol,2021,279:114445.
[45] SUBRAMANIAM S,WEN X Y,JING P.One-step microwave curingdehydration of Gastrodia elata Blume:Relationship between phytochemicals,water states and morphometric characteristics[J].Food Chem,2020,319:126635.
[46] 李晶晶,李君翔,李媛媛,等.植物乳植杆菌发酵天麻酶解液的工艺优化及活性成分分析[J].食品工业科技,2025,46(17):260-271.
[47] YING Z M,SUI G Y,JIA L Q,et al.Simultaneous determination of three compounds in rat plasma by UHPLC-QQQ-MS/MS and its application to pharmacokinetics of Banxia Baizhu Tianma Tang[J]. J Anal Methods Chem,2023,1234:123456.
[48] 陈来凤,倪琳钰,邓成林,等.乳酸菌发酵天麻口服液工艺优化[J].食品工业科技,2023,44(15):193-202.
[49] FISHBACK C,YUAN R,XU L,et al.Substrate tunnel engineering of a type I β-glucosidase for enhanced catalytic efficiency toward gastrodin[J].ACS Catal,2022,12(14):8765-8776.
[50] LI J,ZHANG H,WANG F,et al.Recent advances in beta-glucosidase from Aspergillus niger:Structure,function and industrial applications[J].Bioresour Technol,2022,361:127834.
[51] ZHANG Y,WANG K,LIU T,et al.Advances in Bacillus subtilis expression systems for industrial enzyme production[J].Appl Microbiol Biotechnol,2023,107(6):2457-2474.
[52] CHEN X,WU M,LIU R,et al.Substrate specificity and engineering of type III beta-glucosidases: Structure-function relationships[J]. J Mol Catal B-enzym,2022,194:122365.
[53] WANG S,LI H,ZHANG Q,et al.Gut microbial beta-glucosidases:Roles in phytochemical metabolism and human health[J]. Trends Food Sci Tech,2023,132:251-264.
[54] ZHANG Y,WANG L,ZHAO Y,et al.Structural basis and engineering of esterases for phenolic compound biotransformation[J]. ACS Catal,2023,13(18):10985-11001.
[55] SIRILUN S,CHAIYASUT C,KANTACHOTE D,et al.Functional properties of β-glucosidase-producing Lactobacillus plantarum SC 359 isolated from Thai fermented soybean food[J].LWT-Food Sci Technol,2023,170:113692.
[56] WANG Q,CHEN L,LIU Y,et al.A phenolic esterase from Rhizopus oryzae enables direct conversion of parishin A to bioactive HBA[J].Appl Microbiol Biotechnol,2022,106(15):5189-5201.
[57] LEE F H,WAN S Y,FOO H L,et al.Comparative study of extracellular proteolytic,cellulolytic,and hemicellulolytic enzyme activities and biotransformation of palm kernel cake biomass by lactic acid bacteria isolated from Malaysian foods[J].Int J Mol Sci,2019,20(20):4979.
[58] BEHERA S S,et al.Lactobacillus plantarum with functional properties: an approach to increase safety and shelf-life of fermented foods[J].Trends Food Sci Tech,2024,143:189-203.
[59] 檀茜倩,程笑笑,崔方超,等.泡菜中产β-葡萄糖苷酶植物乳杆菌的益生和酶学特性[J].中国食品学报,2024,24(9):69-79.
[60] CRISTAN A,OSCAR M H,ERIKA Y R,et al.Author response for"production of β-glucosidase by Aspergillus niger CDBB-H-175 on submerged fermentation"[J].Can J Chem Eng,2022,100(7):1489-1501.
[61] MEI J F,CHEN X,LIU J H,et al.A biotransformation process for production of genistein from sophoricoside by a strain of Rhizopus oryza[J].Sci Rep,2019,9(1):6564.
[62] LANGA C N.Characterisation of recombinant beta-glucosidases from Thermomyces lanuginosus SSBP and investigation of their synergistic potential in cellulose hydrolysis[D].Durban:Durban University of Technology,2020.
[63] ZHENG F L,YANG R F,CAO Y L,et al.Engineering Trichoderma reesei for hyperproduction of cellulases on glucose to efficiently saccharify pretreated corncobs[J].J Agr Food Chem,2020,68(45):12671-12682.
[64] KIM S H,YEHUALA G A,BANG W Y,et al.Safety evaluation of Bacillus subtilis IDCC1101,newly isolated from Cheonggukjang,for industrial applications[J].Microorganisms,2022,10(12):2494.
[65] EL-SHAZLY ASMAA I,EL-DEEN A M N,ABDELWAHED N A M,et al.Medium optimization for isoflavone aglycones production from fermented defatted soybean flour by display of β-glucosidase from Bacillus licheniformis NRC24 and evaluation of their antiviral and antitumor activities[J].J Microbiol Biotechn Food Sci,2022,12(2):e5713.
[66] XU H,XIONG A S,ZHAO W,et al.Characterization of a glucose-,xylose-,sucrose-,and D-galactose-stimulated β-glucosidase from the alkalophilic bacterium Bacillus halodurans C-125[J].Curr Microbiol,2011,62:833-839.
[67] SON S H,JEON H L,YANG S J,et al.Probiotic lactic acid bacteria isolated from traditional Korean fermented foods based on β-glucosidase activity[J].Food Sci Biotechnol,2018,27(1):123-129.
[68] SU M,HU Y,CUI Y,et al.Production of β-glucosidase from okara fermentation using Kluyveromyces marxianus[J].J Food Sci Tech,2020,58(1):366-376.
[69] GARCÍA M,LÓPEZ F,RUIZ P,et al.Comparative study of recombinant lipases from different expression systems:Pichia pastoris vs bacterial hosts[J].J Mol Catal B-Enzym,2022,195:111892.
[70] VERMELHO A B,SUPURAN C T,GUISAN J M,et al.Microbial enzyme:Applications in industry and in bioremediation[J]. Enzyme Res,2012,2012:980681.
[71] MARIA G,MARIA D,FRANCISO V.Comparison of the biochemical properties of a recombinant lipase extract from Rhizopus oryzae expressed in Pichia pastoris with a native extract[J]. Biochem Eng J,2011,54(2):117-123.
[72] BRACCO P,MIDDEN N V,ARANGO E,et al.Bacillus subtilis lipase A-lipase or esterase?[J].Catalysts,2020,10(3):308.
[73] TANZI L,ROBESCU M S,MARZATICO S,et al.Developing a library of mannose-based mono-and disaccharides: A general chemoenzymatic approach to monohydroxylated building blocks[J].Molecules,2020,25(23):5764.
[74] EMINE D,MERIH K.Lipolytic activity of lactic acid bacteria isolated from Turkish pastirma[J].Anadolu Univ J Sci Technol C Life Sci Biotechn,2018,7(1):12-19.
[75] DESIREE R N,MICHAEL C,ANNE T.Superior esterolytic activity in environmental Lactococcus lactis strains is linked to the presence of the SGNH hydrolase family of esterases[J]. JDS Communications,2020,1:25-28.